
- This event has passed.
Conference Pr Yohei Shimizu

Pr Yohei Shimizu
Institute for Chemical Reaction Design and Discovery – Department of Chemistry, Hokkaido University (Japan)
Email: shimizu-y@sci.hokudai.ac.jp, +81(11)-706-2719
Catalytic Asymmetric Reactions Empowered by Multiple Non-covalent Interactions between Ligands and Substrates
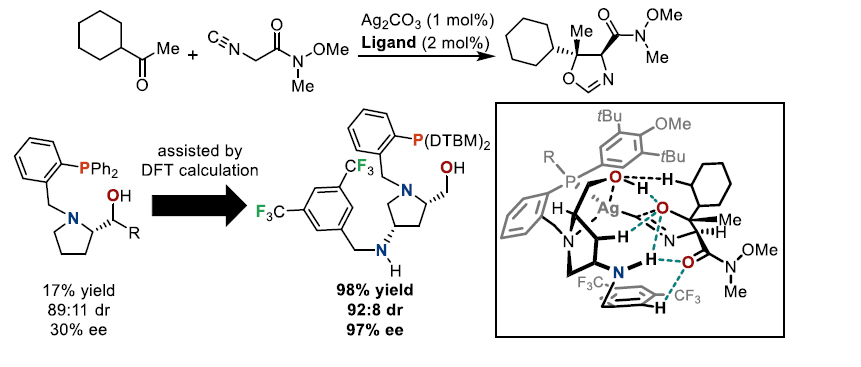
Asymmetric transition metal-catalyzed reactions have been evolved by the development of novel chiral ligands. The creation of a chiral environment in the reaction field by the ligand, and how it controls the approach direction and conformation of the substrate, is crucial for achieving high stereoselectivity. Traditionally, steric repulsion has been regarded as the primary factor governing such control. In contrast, we have focused on developing catalytic reactions that also emphasize attractive non-covalent interactions between the ligand and the substrate. These interactions, such as hydrogen bonding, are actively utilized to control the positioning and conformation of the substrate. As a result, the reaction proceeds not only with high stereoselectivity, but also with high reactivity, allowing the application to less reactive substrates.
In this presentation, I will introduce our recent work on asymmetric copper and silver catalysis using chiral prolinol–phosphine ligands, including analysis by DFT calculations to acquire knowledge on the reaction pathways.1 In particular, I will discuss in detail a copper-catalyzed asymmetric aldol reaction employing sterically demanding dialkyl ketones as electrophiles.2 Insights obtained from DFT calculations were fed back into ligand design, leading to the development of a catalyst system that proceeds with high yields and excellent stereoselectivity.
Références
1. (a) Sakai, S.; Fujioka, A.; Imai, K.; Uchiyama, K.; Shimizu, Y.; Higashida, K.; Sawamura, M. Adv. Synth. Catal. 2022, 364, 2333.
(b) Murayama, H.; Heike, Y.; Higashida, K.; Shimizu, Y.; Yodsin, N.; Wongnongwa, Y.; Jungsuttiwong, S.; Mori, S.; Sawamura, M. Adv. Synth. Catal. 2020, 362, 4655.
(c) Imai, K.; Takayama, H.; Murayama, H., Ohmiya, H.; Shimizu, Y.; Sawamura, M. Org. Lett. 2019, 21, 1717.
