LCC
Engineering first-order spin-orbit coupling in a pentagonal bipyramidal Fe(II) complex and subsequent SMM behavior
Engineering first-order spin–orbit coupling in a pentagonal bipyramidal Fe(ii) complex and subsequent SMM behavior.
Bretosh, V. Béreau, F. Heully-Alary, N. Suaud, C. Duhayon, E. Duverger-Nédellec, N. Guihéry, J.-P. Sutter.
Inorg. Chem. Front., 2025, 12, 9, 3456-3468.
https://doi.org/10.1039/D4QI03255A
Contact:
Jean-Pascal Sutter jean-pascal.sutter@lcc-toulouse.fr
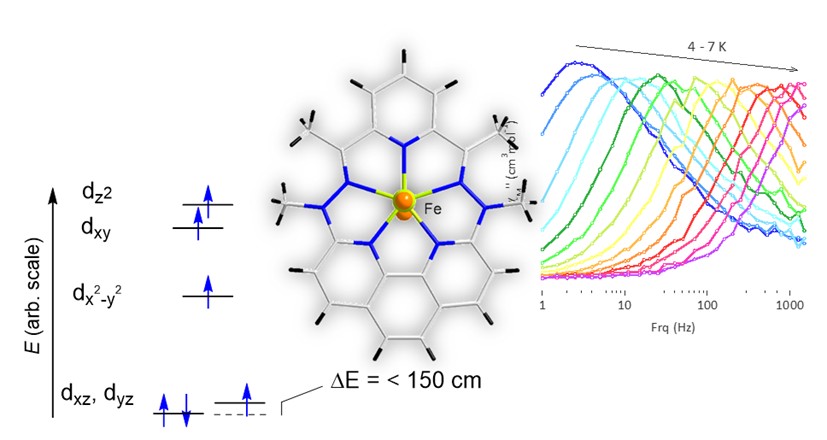
Unlike lanthanide ions, 1st-order spin-orbit coupling (SOC) is cancelled by the ligand field for transition metal ions, and achieving high magnetic anisotropy is generally considered the Holy Grail for these ions. Results obtained with Fe(II) show that a contribution from first-order SOC coupling is not confined to low-coordination number complexes but can be implemented in a pentagonal bipyramid complexes (figure) by proper chemical design. This is confirmed by theoretical calculations and experimental magnetic behaviors consistent with the existence of large magnetic anisotropy, most salient feature being the SMM behaviors with large energy barriers for magnetization reversal. The observation of such a behavior without an applied static magnetic field is quite unique for a mononuclear 3d complex.
LCC
Laboratoire de chimie de coordination du CNRS
205 route de Narbonne, BP 44099
31077 Toulouse cedex 4
France




