LCC
Functionalization and sorption of microporous supramolecular network
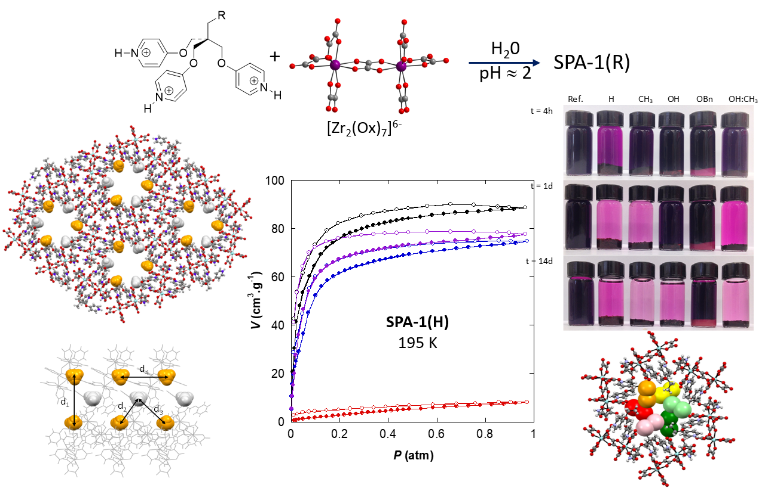
©Sutter Jean-Pascal & Roques Nans/LCC Toulouse
Figure. (Top) H-bonds donnor carrying R groups (H, CH3, OH or OCH2Ph) and H-bonds acceptor implied in formation of SPA-1(R), with single crystal structure of SPA-1(CH3), (down) I2 absorption (from diode solution in hexane) by SPA-1(R) as a function of R group (photos realised after 4h, 1 and 14 days) and single crystal structure of SPA-1(CH3) after adsorption of I2.
Microporous solids formed from molecules associated by hydrogen bonds (H-bonds) are a class of molecular materials of growing interest. Typical properties of porous solids, such as permanent porosity, selective adsorption and sequestration of adsorbates or even detection, have been obtained for such supramolecular architectures.
Our approach to develop such materials involves negatively charged coordination complexes, typically metal-oxalate complexes, and pyridinium-type organic cations. The former act as H-bond acceptors and the latter as H-bond donors. The resulting ionic H-bonds are stronger than classical H-bonds. This particularity, which makes our approach original, has allowed access to new series of iso-structural porous structures called SPA-1(R), whose channels are decorated by polar, apolar R groups or a mixture of both (see figure). These modifications has allowed adjustment of sorption characteristics for the solids while maintaining very similar porosities. As an illustration, for R = CH3, rapid adsorptions of carbon dioxide or iodine are obtained whereas they are much less efficient (or even non-existent) when R = OH. However, this last group is ideal for rapid water adsorption at low saturating vapor values and for sequestering its adsorbate more strongly.
These exceptional results in the field of porous supramolecular architectures are linked to the robustness of the synthons association involved in the formation of SPA-1(R), forcing an identical structure for each SPA-1(R) independently of the R group carried by the cation.
LCC CNRS
Laboratoire de chimie de coordination du CNRS
205 route de Narbonne, BP 44099
31077 Toulouse cedex 4
France




